2026 Swiss TB Award
The SwissTB award has been given this year to M. Tizian Griesser.
Selective Inhibition of M. tuberculosis GpsI: A Novel Strategy Targeting RNA Metabolism

As multidrug-resistant tuberculosis (MDR-TB) continues to pose a severe global health threat, the discovery of innovative drug targets with unique mechanisms of action is paramount. At the Sander Lab at the Institute of Medical Microbiology (IMM), University of Zurich, we focus on finding new therapeutic agents and targets. In our recent study, published in Nucleic acid Research (2025), we identified the mycobacterial PNPase GpsI as an essential and previously underexplored therapeutic target.
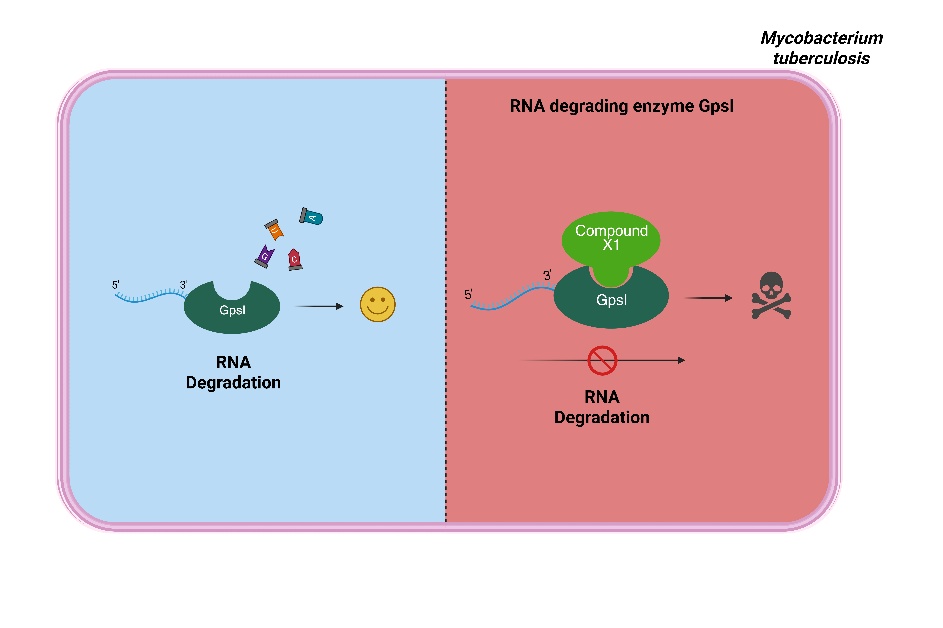 Figure : X1 targets the previously underexplored and essential GpsI enzyme.
Figure : X1 targets the previously underexplored and essential GpsI enzyme.
Key Findings of the Publication:
- Target Identification: Our team demonstrated that GpsI (Rv2783c) is essential for Mycobacterium tuberculosis (Mtb) growth, as it plays a critical role within the RNA degradosome to regulate mRNA turnover.
- Discovery of a Potent Inhibitor: Through high-throughput whole-cell screening, we identified the small molecule X1, which exhibits potent bactericidal activity against Mtb in the low micromolar range and targeting GpsI.
- Structural Insights via Cryo-EM: In collaboration with the Dubochet Center Lausanne, we utilized high-resolution cryo-electron microscopy to visualize the ternary GpsI-X1-RNA complex. This revealed that X1 acts as a noncompetitive inhibitor, binding with high affinity to the enzyme's phosphate-binding site and obstructing RNA access to the catalytic center.
- Molecular Basis of Selectivity: Our research explains the selective specificity of X1 for the Mtb complex. We pinpointed residues 328 and 527 as the molecular "gatekeepers"; natural polymorphisms at these positions in other mycobacteria, such as M. smegmatis, confer innate resistance.
Impact and Outlook: This work by the Sander Group provides a structural and biochemical blueprint for a new class of antibiotics. By targeting the previously untapped pathway of RNA decay, this research opens new avenues for overcoming existing resistance mechanisms and developing highly specific treatments for tuberculosis.
